Executive Summary
The second edition of GAMP 5, published by ISPE (International Society for Pharmaceutical Engineering), represents the most significant update to computerised system validation (CSV) guidance in over a decade. It fundamentally shifts the validation paradigm from a documentation-heavy, prescriptive compliance model to a risk-based, science-driven, lifecycle-oriented approach.
For Pharmaceutical IT leaders, this is not merely a regulatory housekeeping exercise. GAMP 5 Edition 2 redefines how systems are categorised, validated, and maintained, embedding modern software development practices, cloud-readiness, and data-integrity principles into the compliance framework. With industry expectations for full operational alignment by August 2026, the window for preparation is narrow, and the stakes are high.
Key Imperative
Pharma IT organisations that treat GAMP 5 Edition 2 as a documentation update will fall behind. Those that use it as a strategic opportunity to modernise their validation architecture, reduce redundancy, and accelerate system delivery will gain a decisive competitive and regulatory advantage.
This guide is written for Pharmaceutical IT leaders, CSV leads, Quality Assurance professionals, and senior management who need a clear, practical, and comprehensive understanding of what GAMP 5 Edition 2 means for their organisations, what must change, and how to lead that change effectively before the 2026 compliance horizon.
I. What Is GAMP 5 Edition 2?
1.1 Background and Context
GAMP (Good Automated Manufacturing Practice) is the de facto global standard for validating computerised systems used in pharmaceutical manufacturing, laboratory, clinical, and quality operations. It is authored and maintained by ISPE's GAMP Community of Practice and is recognised by major regulators, including the FDA, EMA, MHRA, and PMDA.
The first edition of GAMP 5 was published in 2008 and defined the Software Category framework (Categories 1 through 5) that became the global standard for CSV. While widely adopted, it was increasingly seen as misaligned with modern software development realities, cloud computing, Agile methodologies, SaaS applications, and data integrity as a primary concern were not adequately addressed.
GAMP 5 Second Edition addresses these gaps comprehensively. It is not simply a revision of the 2008 document but a reconceptualisation of how pharmaceutical software validation should be conducted in the modern era.
1.2 Core Philosophy: Risk-Based Thinking
The overarching principle of Edition 2 is that validation effort must be proportionate to the level of risk. This means:
- Higher-risk systems affecting patient safety and data integrity require more rigorous validation.
- Lower-risk systems require lighter-touch approaches.
- Documentation should be purpose-driven and meaningful, not generated for its own sake.
- The focus shifts from producing documents to demonstrating understanding and control of risk.
Paradigm Shift
Edition 2 explicitly discourages 'validation theatre' — the production of voluminous documents that satisfy inspectors but add no real quality value. Regulators increasingly look for evidence of genuine risk understanding, not document counts.
1.3 What Changed from Edition 1

II. The New Software Category Framework
2.1 Moving Away from the 1-5 Model
Edition 2 reorganises software into three broad groups based on intended purpose and configurability. Risk assessment determines the depth of validation and not just the category label alone. This resolves the widespread problem of systems being miscategorised, often deliberately underclassified to reduce validation effort.
2.2 The Three Software Groups
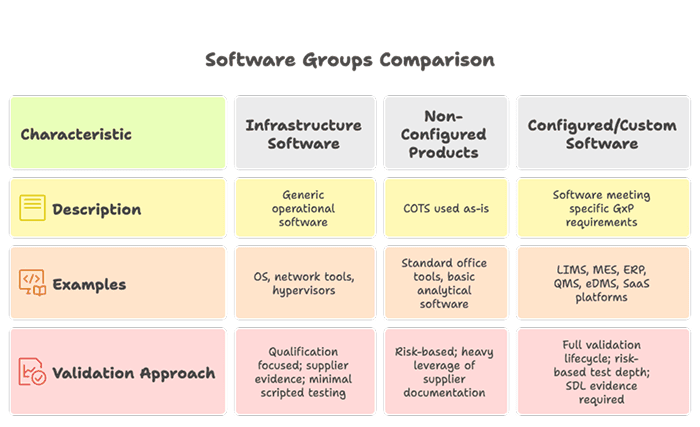
Critical Note for IT Leaders
Most enterprise Pharma IT systems — LIMS, MES, ERP (SAP), CTMS, QMS, eDMS — fall into the Configured/Custom Software group. Validation effort is now explicitly linked to the risk profile of the specific configuration, not just the product category.
2.3 Computer Software Assurance (CSA) Alignment
Edition 2 formally aligns with the FDA's Computer Software Assurance (CSA) framework, emphasising critical thinking over documentation generation. Edition 2 operationalises this by:
- Encouraging teams to define 'sufficient evidence of fitness for intended use' before generating documentation.
- Supporting the use of vendor testing evidence, automated testing outputs, and existing quality management system data.
- Reducing the mandatory creation of test scripts where alternative evidence is adequate.
- Focusing testing effort on critical functionality and high-risk processes.

Understand the Key Differences between CSV & CSA
Read moreIII. Key Themes and Requirements in Edition 2
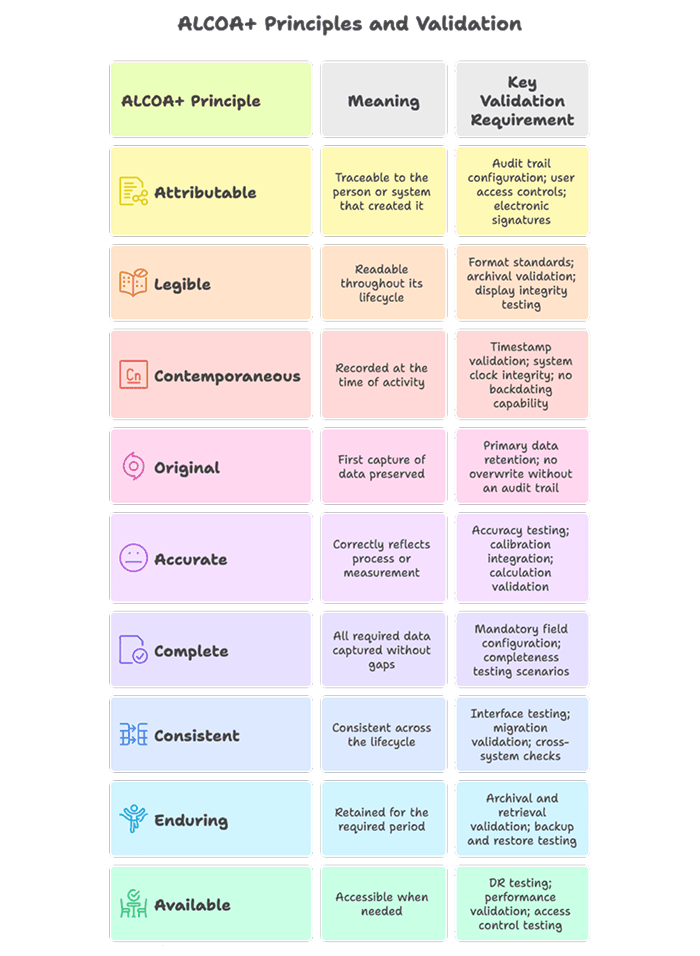
3.1 Data Integrity Now Takes the Center Stage
Data integrity was a background concern in Edition 1. In Edition 2, it is a primary validation requirement. Every computerised system must demonstrate that it maintains data meeting all ALCOA+ principles:
3.2 Agile and Modern Software Development Lifecycles
Edition 2 explicitly recognises that pharmaceutical software is increasingly developed using Agile, DevOps, and CI/CD approaches. Key accommodations include:
- Sprint-based validation activities can be used for configured/custom software.
- Automated testing frameworks (unit, integration, regression) are recognised as valid validation evidence.
- Change management within Agile sprints can be managed via a sprint review rather than traditional change control.
- CI/CD pipelines with appropriate controls satisfy requirements for controlled deployment and release management.
For IT Leaders
If your organisation runs SAP S/4HANA implementations, cloud migrations, or custom builds using Agile, Edition 2 enables you to validate without breaking your delivery cadence. This requires your QA team to be upskilled in Agile validation approaches, a significant capability investment.
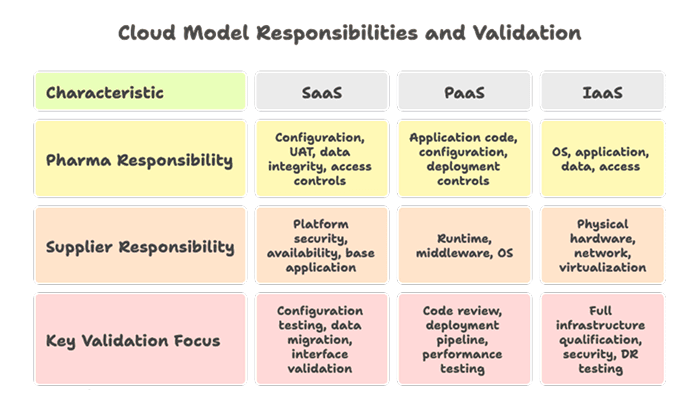
3.3 Cloud and SaaS Validation
Edition 2 provides specific guidance on cloud and SaaS environments, covering:
- Supplier qualification for cloud providers (AWS, Azure, GCP) as infrastructure suppliers.
- Shared responsibility models which clarify what the pharma company must validate vs. what the supplier must evidence.
- Continuous validation approaches for SaaS platforms that update on vendor schedules.
- Data residency, backup, and business continuity as part of the validated system.
- Third-party audit rights and SOC 2 / ISO 27001 as supplementary qualification evidence.
3.4 Supplier Assessment and Management
Edition 2 significantly strengthens supplier management. Validated supplier quality processes can reduce (but not eliminate) the organisation's own validation burden. Key requirements:
- Formal supplier assessment before system selection and not just after contract signing.
- Quality agreements with all GxP software suppliers, including SaaS vendors.
- Ongoing supplier monitoring and not just a one-time audit.
- Supplier change notification processes, especially critical for SaaS/cloud vendors who update continuously.
- Right-to-audit clauses in all supplier contracts for GxP-critical systems.
Practical Consideration
Many Pharma IT teams find that existing SaaS vendor contracts lack adequate quality agreement provisions. Reviewing and renegotiating supplier contracts is often one of the first practical actions required for Edition 2 compliance.
3.5 Lifecycle Management and Periodic Review
Edition 2 places greater emphasis on the ongoing maintenance phase. Validated status must be actively maintained through:
- Periodic review of validated systems at risk-based frequency — typically annual to triennial.
- Structured change control for all GxP-impactful changes.
- Retirement and decommissioning procedures that preserve data integrity.
- Legacy system management strategies for systems that cannot be easily updated.
IV. The August 2026 Compliance Horizon
4.1 Why August 2026 Matters
While GAMP 5 Edition 2 was published in 2022, the pharmaceutical industry operates on extended adoption timelines due to the complexity of existing validated system portfolios and ongoing regulatory cycles. August 2026 has emerged as the de facto industry target based on:
- Regulatory inspection readiness: FDA, EMA, and MHRA are increasingly referencing Edition 2 principles in observations and warning letters.
- Industry consortium commitments: major pharmaceutical trade bodies and ISPE chapters have signalled August 2026 as the readiness benchmark.
- Supplier roadmaps: major GxP software vendors (Veeva, SAP, LabWare, Sparta Systems) are aligning their documentation to Edition 2 by this timeframe.
- New system validation: any new GxP system implemented from mid-2025 onward should be validated to Edition 2 standards; existing systems require gap assessments.
Regulatory Signal
FDA 483 observations and warning letters increasingly reference 'inadequate data integrity controls' and 'insufficient risk assessment' — hallmarks of Edition 1-era approaches. Inspectors are not yet mandating Edition 2 but are clearly inspecting against its principles.
V. Key Challenges for Pharma IT
5.1 The Scale of the Existing Portfolio
Most established pharmaceutical companies have validated system portfolios comprising dozens to hundreds of GxP-critical applications. Re-assessing and remediating this portfolio while running day-to-day IT operations is the central execution challenge:
- Large companies may have 200-500+ validated systems requiring gap assessment.
- Validation documentation for many systems dates back 10-15 years.
- Legacy systems may lack the technical capability to meet Edition 2 data integrity requirements.
- IT teams are typically not sized for a portfolio-wide remediation programme.
5.2 The Knowledge and Skills Gap
Edition 2 introduces concepts that many validation professionals trained under Edition 1 are not yet familiar with:
- Computer Software Assurance (CSA) principles and critical thinking frameworks.
- Agile validation methodologies and sprint-based evidence capture.
- Cloud shared responsibility models and SaaS validation approaches.
- Data integrity by design, moving from retrofitted controls to built-in integrity.
- Risk-based critical thinking, a qualitative skill that documentation-trained teams find genuinely difficult.
Skills Challenge
The shift from 'follow the SOP' to 'apply critical thinking' is not easily trained in a classroom session. It represents a genuine capability transformation that requires investment in coaching, mentoring, and practice, not just training courses.

How To Write a CAPA Report That Stands Up To Any Audit
Read more5.3 SaaS and Cloud Complexity
Many SaaS-delivered GxP systems were implemented under Edition 1 frameworks (or no framework at all), resulting in:
- No quality agreements in place with SaaS vendors.
- No supplier change notification processes — updates apply automatically without validation review.
- Insufficient testing of cloud-to-on-premise interfaces and data flows.
- Data residency and backup validation are not addressed.
- Business continuity is not part of the validated system definition.
5.4 Agile/DevOps Integration
While Edition 2 accommodates Agile, integrating validation into Agile delivery is not straightforward. Common failure modes include:
- QA review added as a gate after sprint completion, breaking Agile cadence.
- Validation documentation written retroactively, negating the contemporaneous requirement.
- Automated test evidence is not structured to satisfy GxP audit requirements.
- Change control processes are too slow for CI/CD deployment frequencies.
5.5 Resource and Budget Constraints
Edition 2 compliance requires dedicated resources that most Pharma IT budgets have not explicitly planned for:
- Specialist validation consultants and CSV leads are in high demand and short supply globally.
- Edition 2 remediation competes with ongoing transformation projects for skilled resources.
- Budget holders may not yet appreciate the regulatory risk of non-compliance.
- ROI of compliance activities is difficult to quantify, making budget approval challenging.
VI. Solutions and Implementation Strategies
6.1 Start with a Portfolio-Level Gap Assessment
The first critical action is understanding your current state. A structured, risk-stratified gap assessment should be completed before any remediation begins:
6.2 Update the Framework Before the Systems
A common mistake is attempting system-level remediation before the organisational framework is updated. The SOP and procedural framework must come first:
- Update the Validation Master Plan (VMP) to reflect Edition 2 principles and philosophy.
- Update the CSV SOP suite, categorisation, risk assessment, testing, change control, periodic review.
- Update templates, URS, Validation Plan, Test Scripts, Validation Summary Report.
- Publish interim guidance for validation teams on Edition 2 key changes.
- Train all CSV-involved staff before system-level remediation begins.
6.3 Adopt a Risk-Based Remediation Approach
Not all systems need the same level of remediation effort. For each system, the remediation decision should consider:
- What is the impact on patient safety and product quality if this system fails?
- What is the data integrity risk profile of the system?
- Is the system approaching end-of-life, or is it a long-term platform?
- What is the cost of remediation vs. the cost of non-compliance?
Strategic Principle
The goal is not to re-validate every system. The goal is to ensure every GxP system has a risk-appropriate validation package reflecting its current configuration and use, and that you can demonstrate genuine understanding and control of the associated risks.
6.4 Build an Edition 2-Native Validation Toolkit
Build a new toolkit from the ground up that operationalises Edition 2 by design:
- Risk Assessment Templates using the Edition 2 risk matrix framework.
- Supplier Qualification Questionnaires covering SaaS, cloud, and traditional suppliers.
- Cloud Validation Protocol Templates for SaaS, PaaS, and IaaS deployment models.
- Agile Validation Evidence Templates, sprint-based evidence capture forms.
- Data Integrity Assessment Checklists, ALCOA+ structured review tools.
- Critical Thinking Decision Trees for categorisation, testing depth, and documentation scope.
6.5 Engage Suppliers Early
For SaaS and cloud-based GxP systems, supplier engagement is a critical path item:
- Inventory all GxP SaaS and cloud systems and their current quality agreement status.
- Issue supplier questionnaires aligned to Edition 2 requirements.
- Negotiate and execute Edition 2-compliant quality agreements, including change notification obligations.
- Request and review supplier validation documentation packages and test evidence.
- Establish ongoing supplier monitoring, at minimum annual review.
Contract Note
Supplier quality agreements for SaaS platforms must specifically address: change notification lead times, access to validation documentation, audit rights, data residency, backup and recovery, and business continuity. Generic IT service agreements do not meet the requirements of Edition 2.
6.6 Integrate Validation into Agile Delivery
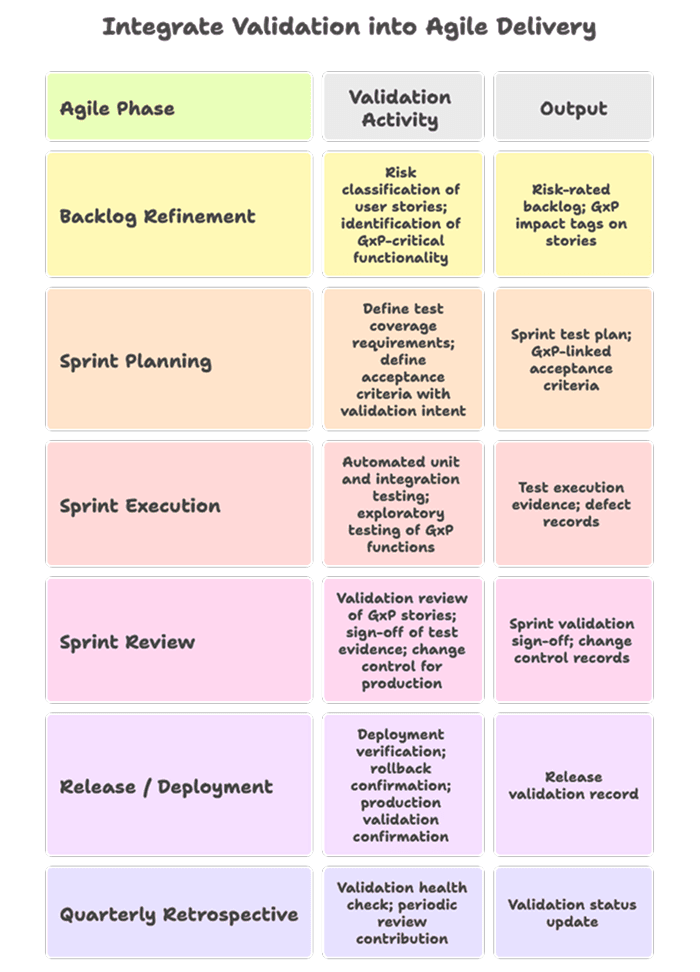
VII. What Pharma IT Leaders Should Look For
7.1 Readiness Scorecard
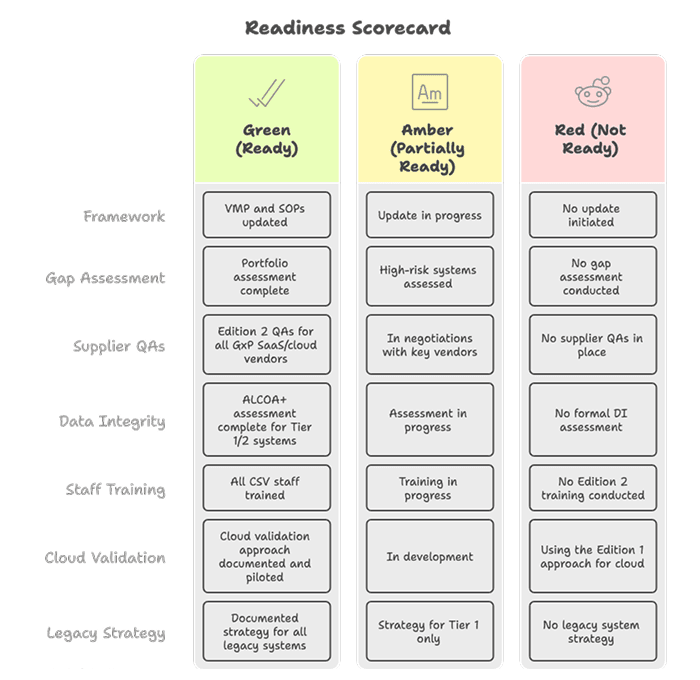
7.2 New Technology Partners and Vendors
When evaluating new GxP software vendors, Edition 2 compliance capability should be a selection criterion. Look for:
- Vendor-maintained validation documentation packages aligned to Edition 2.
- Established change notification and communication processes for regulated customers.
- Evidence of SDL maturity — SDLC documentation, automated testing, code review processes.
- SOC 2 Type II, ISO 27001, or equivalent certifications.
- Experience with SaaS validation in regulated pharmaceutical environments.
- Willingness to enter quality agreements with appropriate provisions.
7.3 Regulatory Inspections
Inspectors are increasingly assessing systems against Edition 2 principles. Prepare your teams for:
- Questions about how systems are risk-classified and what that means for validation depth.
- Requests to see evidence of data integrity controls — audit trails, access controls, electronic signatures.
- Scrutiny of supplier qualification evidence for cloud and SaaS systems.
- Questions about how changes are managed for continuously-updating SaaS platforms.
- Requests for periodic review records demonstrating a maintained validated status.
Inspection Readiness Tip
Inspectors increasingly look for a concise reference that maps each GxP system to its validation status, risk classification, data integrity controls, and last periodic review date. This should be prepared and maintained as a living document.
7.4 Emerging Technologies
Edition 2 provides early guidance on AI/ML, advanced analytics, and process automation. Leaders should be alert to:
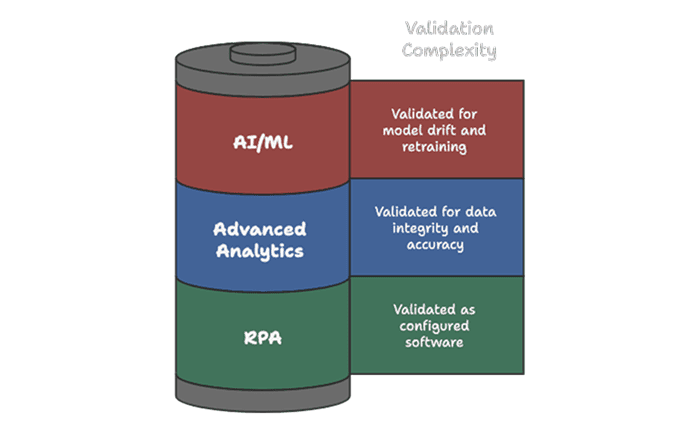
VIII. Building an Edition 2 Implementation Programme
8.1 Recommended Programme Structure

8.2 Governance Model
- Executive Sponsor: typically VP IT or CIO, with QA Director co-sponsorship.
- Programme Steering Committee: IT, QA, and Regulatory Affairs leadership, meeting monthly.
- Dedicated GAMP 5 Programme Manager: with both IT and validation expertise.
- Work Stream Leads: accountable for each work stream delivery.
- CSV Centre of Excellence: technical authority for Edition 2 interpretation and application.
8.3 Key Performance Indicators
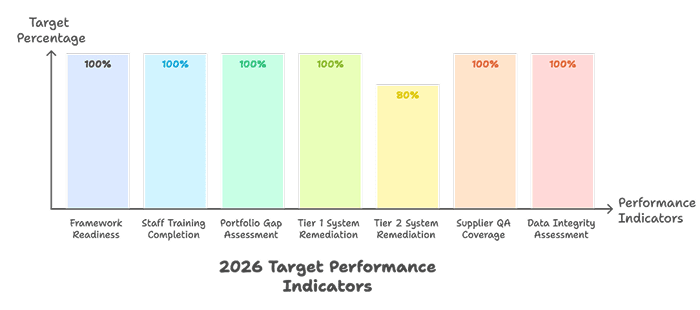
IX. Financial and Resource Planning
9.1 Building the Business Case
Securing budget requires a business case that frames the investment as a strategic enabler, not just a compliance obligation:
- Regulatory Risk Mitigation: FDA warning letters and consent decrees cost significantly more than proactive compliance programmes.
- Operational Efficiency: Edition 2's risk-based approach typically reduces total validation documentation volume by 20-40%, reducing long-term overhead.
- Accelerated System Delivery: Agile-integrated validation reduces time-to-validated status for new systems.
- Audit Readiness: a well-governed programme reduces inspection preparation effort and risk.
- Modernisation Platform: Edition 2 compliance enables confident adoption of cloud, AI, and automation.
9.2 Resourcing Strategy
- Internal CSV team: upskilled and redeployed to framework development and high-value remediation.
- Specialist validation consultancy: for gap assessment methodology, complex remediation, and regulatory interpretation.
- System vendor resources: leveraging vendor validation packages and Edition 2 support services.
- Training providers: accredited ISPE training and certification for CSV staff.
Resource Reality
The global shortage of experienced CSV professionals with Edition 2 expertise means early movers in the talent market have a significant advantage. Organisations that begin building capability now will be better positioned than those that compete for scarce resources in 2025-2026.
X. Strategic Recommendations for Pharma IT Leaders
Recommendation 1: Lead From the Front
GAMP 5 Edition 2 compliance cannot be delegated entirely to the validation team. IT leaders must champion the programme, secure resources, and ensure alignment between IT strategy and compliance requirements. A leadership vacuum will result in fragmented, underfunded, and ultimately ineffective implementation.
Recommendation 2: Make Data Integrity a Design Principle
Do not approach data integrity as a retrofitted control. Require it as a design criterion for every new system acquisition, configuration, and integration. Embed ALCOA+ requirements in vendor selection criteria, design reviews, and UAT sign-off checklists from today forward.
Recommendation 3: Treat Cloud Validation as a First-Class Citizen
Cloud and SaaS validation can no longer be an afterthought. Develop your cloud validation framework now, engage SaaS vendors on quality agreements now, and ensure every cloud-hosted GxP system has a clear, documented, and maintained validation status.
Recommendation 4: Invest in Critical Thinking Capability
The most fundamental shift in Edition 2 is from procedural compliance to risk-based critical thinking. Invest in developing this in your teams through training, mentoring, and psychological safety, so they can exercise and document judgment rather than default to over-documentation.
Recommendation 5: Build a Perpetual Programme, Not a Project
Edition 2 compliance is not a project with a defined end date. It is a perpetual programme of validation lifecycle management. Structure your organisation, processes, and metrics to sustain ongoing compliance, not just achieve a milestone.
Recommendation 6: Engage Regulatory Intelligence Proactively
Track FDA, EMA, and MHRA guidance publications, inspection trends, and warning letter patterns as ongoing intelligence inputs. What regulators cite in 2025 inspections will tell you where to focus validation efforts in 2026.
Recommendation 7: Use Edition 2 as a Modernisation Catalyst
Use Edition 2 implementation as the forcing function to retire legacy systems, standardise platforms, and modernise your validated system portfolio. A compliant, modern portfolio is not just a regulatory asset — it is a business capability asset.
XI. Conclusion
GAMP 5 Second Edition represents a genuine evolution in how the pharmaceutical industry should approach computerised system validation. It is more intellectually demanding than its predecessor, requiring genuine risk-based thinking rather than procedural compliance. It is also more enabling, providing frameworks for cloud validation, Agile delivery, and supplier-leveraged validation that were absent from Edition 1.
For Pharmaceutical IT leaders, the compliance horizon is simultaneously a regulatory imperative and a strategic opportunity. Organisations that approach this as a tick-box exercise will produce compliant documentation but miss the broader opportunity to transform how IT delivers validated systems. Those that approach it as a strategic programme will emerge with more efficient validation processes, more modern system portfolios, and stronger regulatory relationships.
The foundation for success is clear: a structured gap assessment, an updated framework, strong supplier relationships, trained and critically-empowered teams, and leadership that understands why this matters. None of these requires waiting for a regulatory mandate. All of them are available today.
Closing Thought
The best time to begin your GAMP 5 Edition 2 programme was when Edition 2 was published in 2022. The second-best time is today. Every month of delay narrows the window before the end of 2026 and increases the risk of under-resourced, reactive compliance rather than proactive, strategic transformation.
XII. Appendix
Appendix A: Key Terms and Definitions

Appendix B: Key Resources and References
- ISPE GAMP 5: A Risk-Based Approach to Compliant GxP Computerised Systems (Second Edition, 2022).
- FDA Draft Guidance: Computer Software Assurance for Production and Quality System Software (2022).
- EU GMP Annex 11: Computerised Systems (Current version).
- MHRA GxP Data Integrity Guidance (Current version).
- ICH Q9(R1): Quality Risk Management.
- ICH Q10: Pharmaceutical Quality System.
- ISPE GAMP Good Practice Guide: Data Integrity — Key Concepts.
- ISPE GAMP Good Practice Guide: Cloud Computing (Second Edition).
- ISPE GAMP Good Practice Guide: Validation of Laboratory Computerised Systems.
- ISPE GAMP Good Practice Guide: Enabling Innovation (Agile, AI/ML).
