The Butterfly Effect: A New Lens on Women’s Health
- Cover Story – Women’s health, hormones, metabolism, and the expanding female health market.
- Pharma News – Global updates on FDA approvals, clinical trials, partnerships, and industry movements.
- In Talks with Leadership – Featuring insights from Dr. A.R.M. Rao and Dr. Sweta Raghavan on innovation and industry transformation.
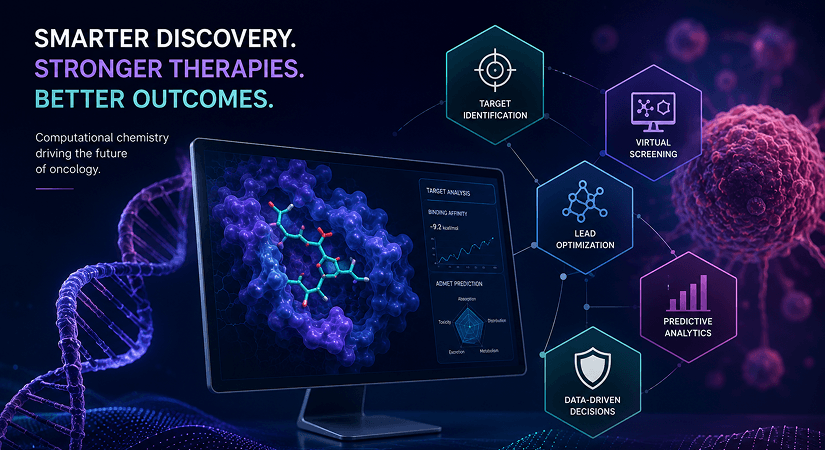
- Market Insights & Innovation – Funding trends, AI in drug development, and India’s innovation ecosystem.
- Marketing, HR & Startup Lens – Pharma marketing challenges, talent strategies, and emerging startup stories.








20260521124126.webp&w=256&q=75&dpl=dpl_BVcZdrQxoXxuGu2R9UbYtTFv4MMh)




















