by Mrudula Kulkarni
7 minutes
How Digital Transformation Is Rewriting the Economics of Drug Development
Digital transformation is reshaping drug development economics by reducing risk, improving R&D efficiency, and enabling smarter pharma decisions.

Drug development has always been expensive.
What is changing today is not the ambition of pharmaceutical innovation, but the economics behind how drugs are developed.
Digital transformation in drug development is reshaping where costs arise, how risks are identified, and how value is created across the pharmaceutical lifecycle.
From early drug discovery to clinical trials, manufacturing, and regulatory submissions, digital technologies are enabling pharma companies to make smarter decisions earlier, reducing late-stage failures and improving R&D efficiency.
This shift is not about replacing scientists with algorithms. It is about augmenting human expertise with data-driven insight, fundamentally altering the cost structure of pharmaceutical R&D.
The Traditional Economics of Drug Development
Historically, the economics of drug development in the pharmaceutical industry have been defined by three persistent challenges:
- High attrition rates during clinical development
- Long development timelines often exceeding 10 to 15 years
- Massive capital investment locked in long before commercialization
Industry data suggests that only one in ten drug candidates entering clinical trials ultimately reaches the market. Late-stage failures are especially costly, consuming years of work and billions of dollars.
In this traditional model, inefficiency was often accepted as inevitable. Digital transformation is challenging that assumption.
Digital Transformation in the Pharmaceutical Industry: A Structural Shift
Digital transformation in the pharmaceutical industry goes far beyond automation or digitizing paperwork. Its true impact lies in reshaping decision-making economics.
Digital technologies enable earlier risk identification, support better portfolio prioritization, improve knowledge retention across teams and programs, and create faster feedback loops that allow pharmaceutical organizations to make timely, data-driven decisions throughout the drug development lifecycle.
By shifting intelligence upstream in the drug development lifecycle, pharmaceutical companies can terminate weak programs earlier, conserve capital, and redirect resources toward high-probability assets. AI in pharma enables earlier portfolio prioritization.
AI and Data-Driven Drug Discovery
Early-stage drug discovery has traditionally relied on time-consuming laboratory experimentation. Today, AI and machine learning in pharma are transforming this phase.
Key applications include:
- AI-driven target identification
- In silico compound screening
- Predictive toxicity and efficacy modeling
By integrating biological data, genomics, and real-world datasets, pharma companies can reduce the number of compounds entering costly preclinical studies.
The economic impact is significant: fewer failed molecules, reduced discovery costs, and faster progression to viable candidates.
Digital Clinical Trials and Cost Optimization
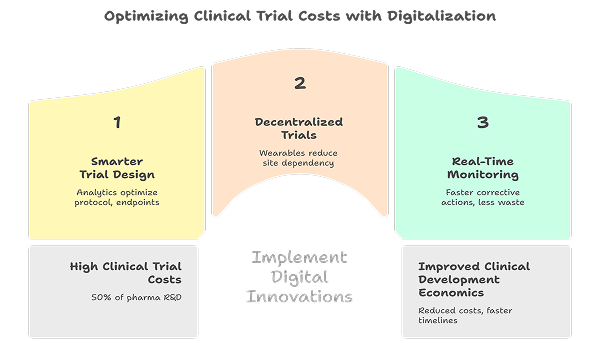
Clinical trials account for nearly 50 percent of total pharmaceutical R&D costs, making them the most economically sensitive stage of drug development.
Smarter Trial Design
Advanced analytics and historical trial data support better protocol design, optimized endpoints, and realistic enrollment strategies, reducing costly amendments and delays.
Decentralized and Hybrid Clinical Trials
Digital clinical trials leverage wearable devices, remote monitoring, and virtual platforms to reduce site dependency, accelerate patient recruitment, and improve retention.
Real-Time Data Monitoring
Digital trial management systems provide real-time visibility into trial performance, enabling faster corrective actions and reducing wasted spend.
Together, these innovations significantly improve the economics of clinical development. Digital tools are improving clinical trial efficiency.
Digital Manufacturing in Pharma: Reducing Variability
Manufacturing failures, batch rejections, and scale-up challenges can quickly erode profitability. Digital transformation in pharmaceutical manufacturing improves predictability and compliance through:
- Digital twins for process simulation
- Advanced process analytics
- Predictive maintenance and quality monitoring
These digital manufacturing tools reduce variability, improve yield, and minimize costly deviations, protecting margins without compromising regulatory compliance.
Regulatory Digitalization and Faster Market Access
Regulatory delays significantly increase development costs and slow patient access to therapies, and digital transformation helps address this by strengthening data integrity and traceability, enabling submission-ready documentation, and supporting real-world evidence generation that aligns more closely with evolving regulatory expectations.
Regulators increasingly expect digital maturity from pharmaceutical companies. Organizations that invest in digital regulatory systems benefit from smoother approvals, fewer queries, and faster time-to-market, directly improving drug development economics. Regulatory digitalization accelerates approvals and reduces rework.
Knowledge Retention as an Economic Advantage
One of the hidden costs in pharmaceutical R&D is knowledge loss. When teams change or projects pause, valuable insights are often lost.
Digital platforms that centralize experimental data, decisions, and outcomes convert institutional knowledge into a reusable asset. This reduces redundant experimentation, prevents repeated mistakes, and strengthens long-term R&D efficiency.
Over time, knowledge retention becomes a powerful economic differentiator. Knowledge management reduces repeated experimentation.
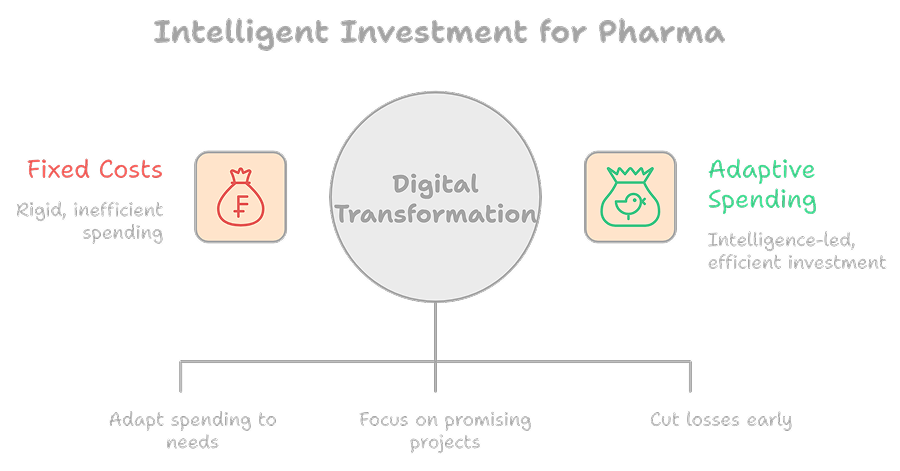
Shifting From Fixed Costs to Intelligent Investment
While digital transformation requires upfront investment, it shifts pharma economics from rigid fixed costs to adaptive, intelligence-led spending.
Pharmaceutical companies can:
- Allocate resources dynamically
- Prioritize high-value assets
- Terminate low-probability programs earlier
This precision does not reduce innovation. It improves capital efficiency across the drug development portfolio.
Cultural Barriers and the Cost of Digital Inaction
The biggest challenge in digital transformation is not technology, but culture. Siloed data ownership, resistance to transparency, and reluctance to trust analytics slow adoption.
However, the economic cost of delaying digital transformation is rising. Companies that resist change face longer timelines, repeated inefficiencies, and reduced competitiveness in an increasingly data-driven pharmaceutical landscape.
The Future of Drug Development Economics
The future of the pharmaceutical industry will not be defined by cheaper drug development, but by smarter economics.
Costs will increasingly shift:
- Earlier in the pipeline
- Toward data infrastructure
- Into predictive capabilities rather than corrective actions
Digital transformation will determine not only how fast drugs are developed, but how sustainably innovation is financed.
Final Thoughts
Digital transformation in drug development is rewriting economics by making complexity visible, measurable, and manageable. In an industry where decisions echo across decades and billions of dollars, data-driven insight is becoming the most valuable asset.
Pharmaceutical companies that embrace this shift are not just accelerating pipelines. They are building resilience into the future of innovation.
FAQs
1. What is digital transformation in drug development?
Digital transformation in drug development refers to the use of data analytics, AI, digital platforms, and connected technologies to improve efficiency, reduce costs, and optimize decision-making across the pharmaceutical lifecycle.
2. How does digital transformation impact drug development economics?
It reduces late-stage failures, shortens timelines, improves resource allocation, and enhances regulatory efficiency, leading to better capital utilization and long-term value creation.
3. Which stage benefits most from digital transformation in pharma?
Clinical development benefits the most, especially through digital clinical trials, improved patient recruitment, and real-time data monitoring.
4. Is digital transformation only for large pharmaceutical companies?
No. Small and mid-sized pharma companies can leverage digital tools to compete efficiently, reduce risk, and optimize limited R&D budgets.
5. What is the biggest barrier to digital transformation in the pharmaceutical industry?
Cultural resistance and siloed data systems are the biggest barriers. Successful transformation requires leadership commitment, cross-functional collaboration, and trust in data-driven decision-making.








